Precision Clinical Development in Dermatology and Ophthalmology
At Precefi, we integrate biomarkers, translational medicine, and digital clinical platforms to improve the efficiency and success rates of clinical trials. By linking biological pathways to measurable clinical outcomes, our approach enables faster proof-of-concept studies, optimized trial design, and more informed development decisions. This strategy helps reduce development risk while accelerating the path toward clinical validation and regulatory approval.
Why Biomarker-
Driven Trials Matter
Many clinical programs fail because the right therapy is tested in the wrong patients, at the wrong dose, or without clear evidence of biological activity.
Biomarkers solve this by:
- Identifying patients most likely to benefit
- Confirming a therapy is engaging its intended target
- Detecting efficacy signals earlier in development
- Enabling smaller, more efficient trials with better patient selection
For sponsors and investors, biomarker-guided development provides scientific clarity, reduces clinical risk, and accelerates key inflection points.
Beyond the Biopsy
Traditional translational research in dermatology is constrained by a fundamental problem: fewer than 20% of eligible subjects consent to optional skin biopsies. Patients resist them, parents refuse them for children, and evolving FDA guidance has further restricted punch biopsy use. The result is inadequate sample sizes, incomplete biomarker datasets, and an inability to track the same lesion longitudinally.
Precefi solves this by embedding proven non-invasive and minimally invasive biomarker collection directly into our site network, managed end-to-end by our translational science team.
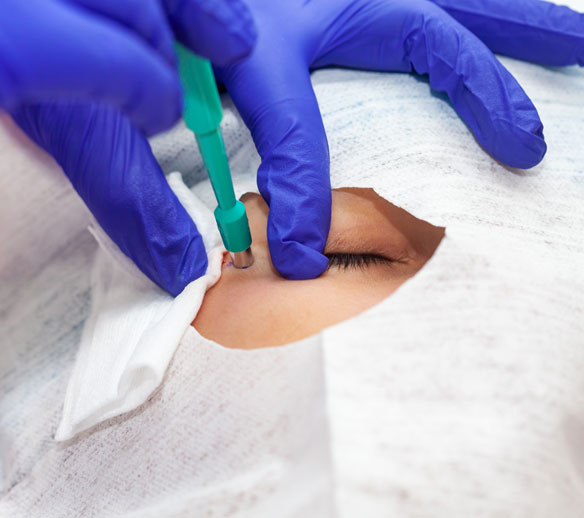
<20%
of eligible subjects consent to optional skin biopsies
FDA Limits
evolving guidance restricts punch biopsy use in clinical trials
Longitudinal
tracking the same lesion over time is nearly impossible with biopsy
Biomarker Collection, Built Into Your Trial
Through established partnerships with best-in-class biomarker collection platforms, Precefi brings cutting-edge skin sampling technology directly to your trial sites as a fully managed service. Our translational science team supports every step, from protocol design through sample logistics, analysis coordination, and data interpretation.
Samples collected via non-invasive or minimally invasive methods support RNA, DNA, and protein analysis across a broad range of indications, processed through a CLIA-certified, CAP-accredited laboratory. The epidermal signal is often cleaner than traditional punch biopsy, which introduces variability through enzymatic tissue separation.
Dermatology
Advances in molecular biomarkers are transforming dermatology drug development. By linking immune and inflammatory pathways to measurable outcomes, biomarker-driven approaches identify patients most likely to respond and confirm target engagement early — enabling more efficient trials and earlier detection of treatment effects.
Ophthalmology
Ophthalmology is uniquely suited for biomarker-driven development, disease processes within the eye can be directly visualized using OCT, retinal imaging, and angiography. Integrating imaging biomarkers with translational medicine enables earlier detection of therapeutic activity, better patient selection, and optimized dosing.
The Precefi Advantage
Precefi’s platform combines clinical research infrastructure, biomarker science, and digital technologies to support more efficient drug development across multiple therapeutic areas.
Add Translational Depth
to Your Next Study
Whether you’re designing a new protocol or adding a biomarker sub-study to an existing trial, Precefi’s translational team will help you select the right collection approach, design a feasible sampling strategy, and execute it consistently across our full site network, in the US and Latin America.

